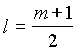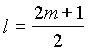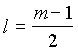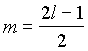SCHOLASTIC APTITUDE TEST 1998
CHEMISTRY
| Time: 1 Hour |
Max.Marks
: 60
|
Instructions:
- Answer must be written either in
English or the medium of instruction of the candidate
in high school.
- There will be no negative marking
- Use of calculators or graph papers
is not permitted
- There are SIX Questions. Answer all
the questions.
I. Multiple choice. Each question has one
or more than one answer(s). Indicate the correct
answer(s) by A,B,C,D. (5x1 = 5 Marks)
1. 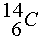 is a beta-active nucleus. A sample 14CH4
gas kept in a closed vessel shows increase in pressure with
time. This is due to
is a beta-active nucleus. A sample 14CH4
gas kept in a closed vessel shows increase in pressure with
time. This is due to
| A) formation of 14NH3
and H2 |
B) formation of 11BH3
and He |
| C) formation of 14C2H4
and H2 |
D) formation of 12CH3,
14NH2 and H2 |
2. For a particular value of azimuthal
quantum number (l) the total number of magnetic quantum
number (m) is given by
3. For a reaction 4A+B ® 2C+2D
which of the following statements is/are
not correct
A) The rate of disappearance of B is one
forth the rate of disappearance of A
B) Rate of appearance of C is one-half the
rate of disappearance of B
C) The rate of formation of D is one-half
the rate of consumption of A
D) Rate of formation of D is one-half the
rate of consumption of A
4. Choose the correct statements
A) Under the similar conditions of pressure
and temperature one mole of
all the gases have the same volumes
B) The average K.E. of gas molecules is
proportional to absolute temperature
C) At absolute zero temperature the volume
of a gas is expected to be zero
D) 2730C and 760 mm of Hg
pressure are the standard temperature and pressure
5. Five valance electrons of 15P
are labelled as
If spin quantum of B and Z is +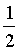 .
.
The group of electrons with three of the
quantum number same are
| A) XYZ, AZ |
B) AB, XYZ, BY |
C) AB |
D) AB, XYZ |
| II. Write the balanced
Chemical Equations for following chemical changes |
(5x2=10 Marks)
|
1. Elemental phosphorus reacts with conc.HNO3.
2. Carbondioxide is passed through a
suspension of lime stone in water.
3. Conc.H2SO4 is drop
wise added to the mixture of NaCl and MnO2.
4. NaCl reacts with an aqueous solution of
NH3 saturated with CO2 gas.
5. Aqueous solution containing potassium
nitrate and ferrous sulphate is treated with conc.H2SO4.
III. Give reasons in one or two sentences
for the following: (10x2 = 20 Marks)
(Write the chemical equations wherever
necessary)
1. The hydroxides of aluminium and Iron are
insoluble in water. However, NaOH is used to separate one
from the other.
2. Helium is monatomic, while hydrogen is
diatomic molecule.
3. Fluorine has less electron affinity than
chlorine but electronegativity of fluorine is more than that
of chlorine.
4. White precipitate is formed when AgNO3
is treated with NaCl(aq) but no precipitate is formed when
AgNO3 is treated with CCl4.
5. Dry SO2 and Cl2
does not bleach dry flowers.
6. Although aluminium is above hydrogen in
the electrochemical series it is stable in air and water.
7. When hydrogen gas is bubbled through
acidified potassium permanganate solution, pink colour is not
discharged, but when few pieces of metal zinc are added to it,
the pink colour disappears immediately.
8. When blue crystals of CuSO4.5H2O
are added to conc.H2SO4 they turn white.
9. Ferric chloride solution used as a
reagent in the laboratory is prepared by dissolving, Ferric
chloride not in mere water, but in hydrochloric acid.
10. Sulphur melts to a clear mobile liquid
at 1190C but on further heating above 160oC
it becomes viscous.
| IV. Fill in the blanks |
(15x1 =
15 Marks)
|
1. The weight of 1x1022
molecules of CuSO4.5H2O is
________________.
(atomic weight of Cu=63.5; S=32;
O=16 amu)
2. The blue complex formed, on addition of
conc.NH4OH solution to a Cu2+ salt
solution has the structure _________________.
3. The temperature at which the real gases
obey the equation pV=nRT under all pressure conditions is
known as ___________.
4. Molten sodium chloride conducts electric
current due to the presence of __________.
5. The atomicity of a species is x and its
atomic weight is y. The molecular weight of the species is
___________________.
6. An element has atomic weight 2Z+6 where
Z is the number of extra nuclear electrons in the atom, then
the number of neutrons in the nucleus is ____________.
7. A container holds an equimolar mixture
of H2 and O2. If through small hole the
mixture is allowed to diffuse, the mass ratio of H2:O2
in the initial diffusing gases is ___________.
8. Helium obtained at STP by the
disintegration of 2 grams of 224Ra after 3.65 days
in a closed vessel t1/2 of 224Ra is 3.65
days _____________.
9. The shift in the equilibrium 2CO + O2
 2CO2
+ Heat
2CO2
+ Heat
on increasing the temperature and
decreasing the volume simultaneously, is __________.
10. Equal weights of hydrogen and methane
are mixed in an empty container at 250C. The
fraction of total pressure exerted by hydrogen is
___________________.
11. An element which can exist as a
positive in acidic solution and also as a negative ion in
basic solution is said to be ____________.
12. A mixture of hydrogen and oxygen (45 ml)
is sparked to form liquid water. The component not in excess
reacts completely and 15 ml is leftover (all measurements are
at the same temperature and pressure). The composition by
volume in the original mixture of H2 and O2
is ____________.
13. The formula of deep red liquid formed
on warming potassium dichromate with KCl in concentrated
sulphuric acid is _______________.
14. The metal which reacts with dilute
nitric acid but becomes passive with conc. Nitric acid is
_______________.
15. Ship hull is protected against
corrosion by attaching zinc blocks because ___________.
V. Arrange the following as stated (5x1
= 5 Marks)
a) Increasing order of ionic size
[N3- , Na+ , F-,
O2-, Mg2+ ]
b) Increasing the strength of hydrogen
bonding (X ……… H - X )
[O, S, F, Cl, N]
c) Increasing order of basic character
[MgO, SrO, K2O, NiO, Cs2O]
d) Increasing order of bond dissociation
[N2, O2, F2,
Cl2]
e) Increasing order of Ist I.P.
[Na, Al, Mg, Si]
VI. Explain the following: (5x1 = 5
Marks)
(Give the Chemical equations wherever required)
1. What do you mean by Hardwater , heavy
water, and distilled water.
2. Name the element whose atomic number is
17 and atomic mass is 35.5. Explain why it has fractional
atomic mass.
3. State and explain the principle which
restricts the number of electrons in an orbital to two.
4. HCl can be prepared by heating NaCl with
conc.H2SO4while HBr cannot be prepared
by heating NaBr with conc.H2SO4.
Explain why?
5. Magnesium metal burns in air to give a
white ash. When this ash is treated with water the odour of
ammonia can be detected. What is the reason.
HOME
![]() is a beta-active nucleus. A sample 14CH4
gas kept in a closed vessel shows increase in pressure with
time. This is due to
is a beta-active nucleus. A sample 14CH4
gas kept in a closed vessel shows increase in pressure with
time. This is due to